South Korea issued the Guidance on Joint Submission Procedure (in Korean) in the middle of August to facilitate potential registrants seeking joint registration. On 22 Sep 2015, the official English version of the Guidance was issued for reference.
Korean domestic enterprises who intend to manufacture or import any of 510 priority existing chemical substances subject to registration (PECs) designated under K-REACH, at a volume of and above 1 ton per year shall jointly submit dossiers within the grace period after identifying other manufacturers or importers of the same chemical substance.
Key steps for preparing a joint submission are outlined as below.
Identify potential registrants
Select a lead registrant
Survey on data availability
Analyze data gap
Purchase or generate additional data
Cost sharing
Prepare and submit dossier
The potential registrants of the same PEC substance can be identified via the Joint Registration Information System. However, to join the system, potential registrants should fill out the following basic information, including
Enterprise name;
Enterprise registration number;
Contact details;
Tonnage band;
Information on manufacture/importation;
Document verifying manufacture/importation of any PECs; and
Information on whether or not registrant wants to be a Lead Registrant
Different legal entities importing/manufacturing PECs should submit corresponding verification documents listed below:
| Type | Essential documents accepted |
| Manufacturer | Certificate of Composition, etc. |
| Importer |
|
| Potential manufacturer or importer |
|
| Only Representative |
|
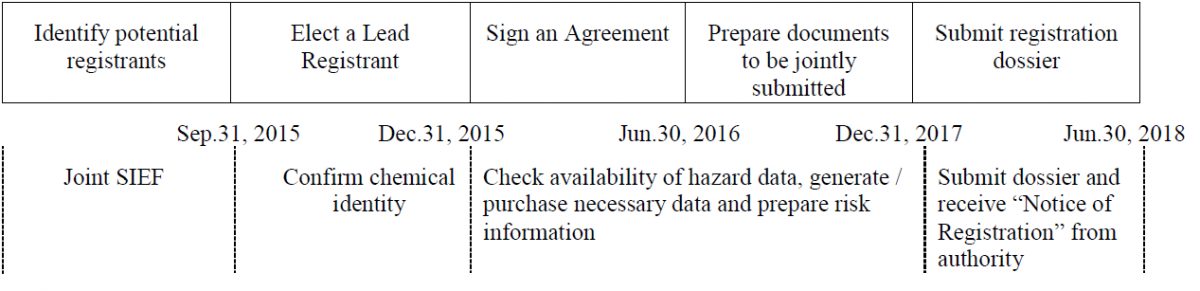
Below is the officially recommended timetable for joint registration of PECs. Potential registrants should take the time to join the system and establish consortium to assure a completion of joint registration when the grace period expires.




 Request a Demo
Request a Demo